Ionic bonds form when two or more ions come together and are held together by. 
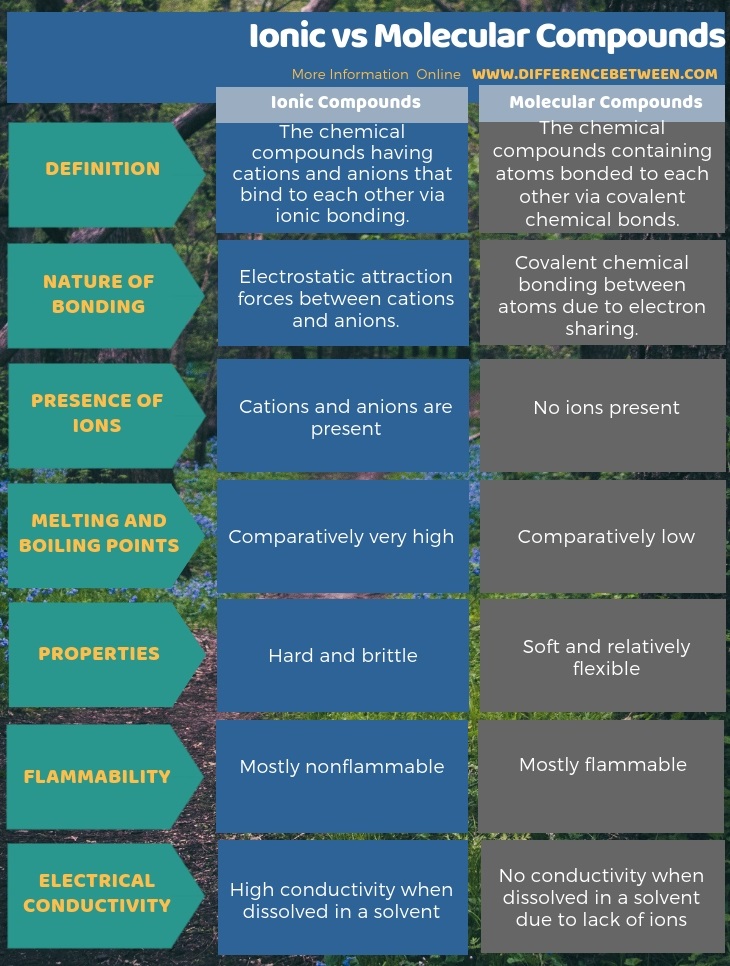
Ionic, on the other hand, refers to substances that are made up of ions, which are atoms or molecules that have gained or lost electrons and have an electric charge. Covalent bonding involves the sharing of electrons between two or more atoms. This results in a cation with 20 protons, 18 electrons, and a 2+ charge. Molecular refers to substances that are made up of molecules, which are groups of atoms held together by covalent bonds. For example, a neutral calcium atom, with 20 protons and 20 electrons, readily loses two electrons. To illustrate, an atom of an alkali metal (group 1) loses one electron and forms a cation with a 1+ charge an alkaline earth metal (group 2) loses two electrons and forms a cation with a 2+ charge, and so on. Unlike ionic bonds, where electrons are either lost or gained by an atom to form charged ions, electrons in covalent compounds are shared between the two atoms. Atoms of many main-group metals lose enough electrons to leave them with the same number of electrons as an atom of the preceding noble gas. You can use the periodic table to predict whether an atom will form an anion or a cation, and you can often predict the charge of the resulting ion. Writing and balancing net ionic equations can be challenging but if you carefully follow the correct steps you will be successful. (b) A sodium cation (Na+) has lost an electron, so it has one more proton (11) than electrons (10), giving it an overall positive charge, signified by a superscripted plus sign. Compounds can be either ionic compounds or molecular. (a) A sodium atom (Na) has equal numbers of protons and electrons (11) and is uncharged. To make sense of these properties, we must recall that molecular compounds consists of independent units called molecules.These molecules are formed when two or more atoms combine by sharing electrons. Atoms of different elements join together in definite proportions to form molecules of compounds. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
